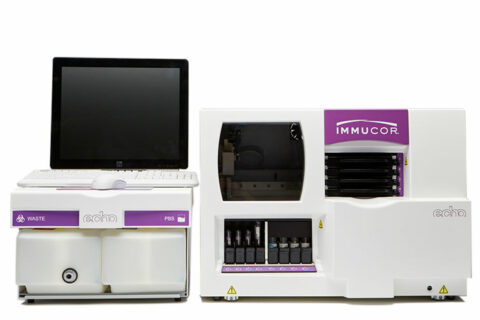
NEO Iris® Benefits
Automated
Throughput
Accuracy
Workflow
The Details
Increase Your Productivity.
NEO Iris® is Werfen’s most productive, fully-automated blood bank instrument. Improve your day-to-day efficiencies with the highest type and screen throughput in the market.
- Dynamic workflow and resource handling
- Add samples at any time with true continuous access
- Remarkable flexibility for STAT priority functionality
- Reflex testing and customized profiles
Flexibility to Optimize Your Lab’s Workflow.
With linear sample and reagent racks for true continuous access, STAT requests never wait. NEO Iris’s dynamic workflow management tool with STAT priority allows you to run any test, in any order, at any time.
High Accuracy.
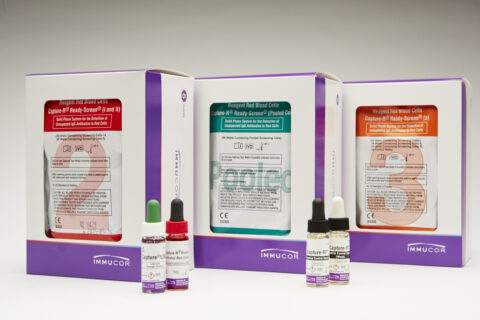
Capture®, Werfen’s IgG specific technology, provides high sensitivity for clinically significant antibodies, without the noise of IgM antibodies.
- Efficient antibody identification with 3 different pre-coated panels, each featuring 14 unique donors
- Real-time test results with flexible views
Quality Control
A laboratory is driven by the quality of the test results it generates. Werfen provides an array of Quality Control reagents designed to meet stringent quality and compliance standards.
| Product | Use |
|---|---|
| corQC® EXTEND Standard | Daily quality control of routine Rh and Kell reagents on the NEO Iris®/NEO® v2.0 |
| corQC® EXTEND Complete | Daily quality control of routine Rh and Kell reagents on the NEO Iris®/NEO® v2.0 |
| DAT Positive Control Cell | DAT Positive antiglobulin control on the Echo Lumena®/Galileo Echo® (v2.0) and NEO Iris®/NEO® v2.0 |
Remote Support
Werfen offers highly secure and compliant remote support to customers across the globe.
Werfen’s remote diagnostics includes the use of state of the art technology, robust processes and adherence to strict policies when providing technical and field support for our instruments.
Our objective is to resolve your technical issue as quickly as possible while ensuring that any exposure to sensitive data is kept to a minimum.
Validation Support
Werfen recognizes sourcing specimens with positive antibodies is challenging. Let us ease the burden of finding positive samples or contriving samples with our Validation Panel:
- Panel of 8 known ready to use samples prepared from human IgG monoclonal antibodies
- Unique barcode identifiers for each sample
Werfen is committed to the immunohematology laboratory and will be your partner from install through validation and go-live.
Product offerings may differ by region, please check with your local Werfen representative for regulatory status in your area.
NEO Iris® Videos
NEO Iris® Downloads

Resource Library
Our Certifications
Experience Our Virtual Lab
Related Products
- Testimonial
- Transfusion
- Customer
Alexis Crisp, MLS (ASCP) CMSBB, Blood Bank Supervisor, Avera McKennan Hospital and University Health Center
Let's Get Started On Your Story- Testimonial
- Transfusion
- Customer
Kerry Dowling, Blood Transfusion Laboratory Manager, Southampton General Hospital University Hospital Southampton NHS Foundation Trust, United Kingdom
Let's Get Started On Your Story- Testimonial
- Transfusion
- Customer
Lisa Knapp, MT(ASCP), Blood Bank Coordinator, Bozeman Health Deaconess Hospital
Let's Get Started On Your Story- Testimonial
- Transfusion
- Customer
Don-Dolcetto Ngilisho, Laboratory Services Officer, Tanzania National Blood Transfusion Service
Let's Get Started On Your Story- Testimonial
- Transfusion
- Customer
Kevin Elman, Transfusion Services Coordinator, North Colorado Medical Center, USA
Let's Get Started On Your Story- Testimonial
- Transfusion
- Customer
Raeann Thomas, Blood Bank Technical Supervisor, Hospital of the University of Pennsylvania, USA
Let's Get Started On Your StoryUpcoming Events,
Webinars & Workshops
Keep Up With Us