Automated Capture-R® Ready-Screen®
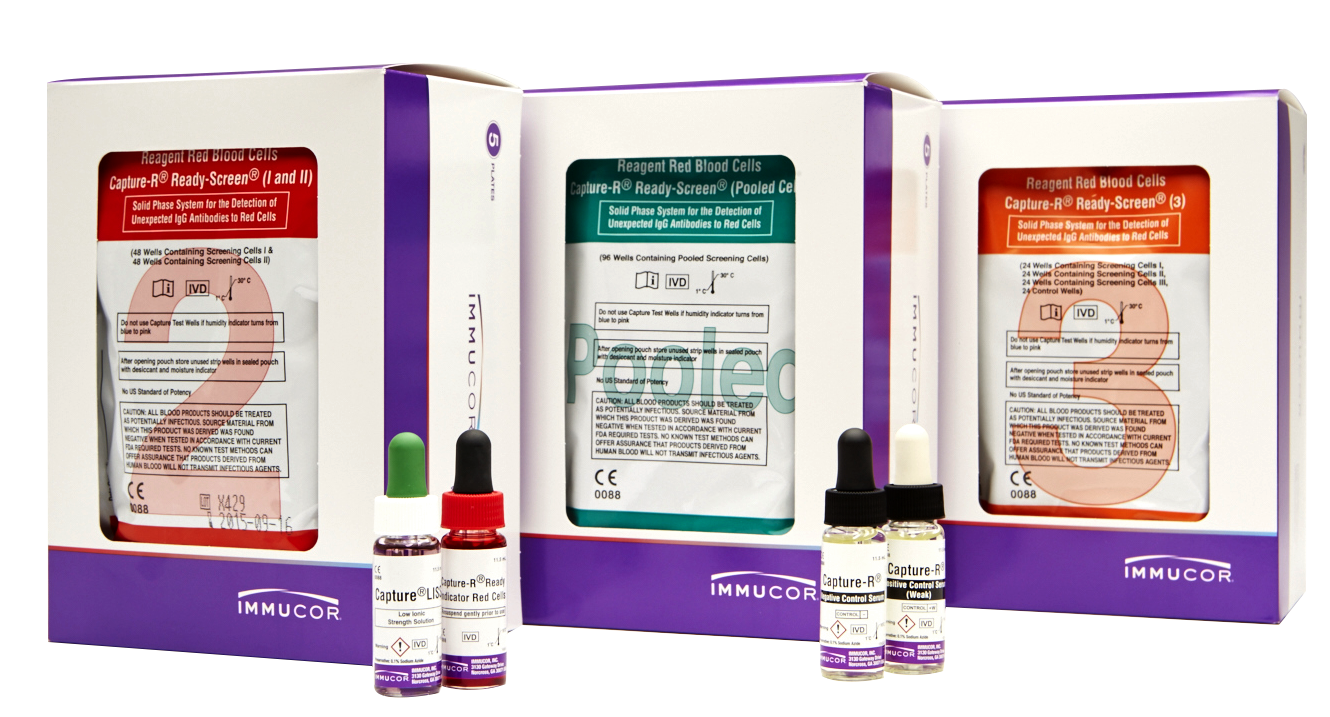
Automated Capture-R® Ready-Screen® Benefits
to Use
Specificity
Flexibility
The Details
Take your laboratory to the next level of productivity with Capture®, a proven IgG specific solid phase technology for antibody screening.
High Accuracy.
- Detect clinically significant antibodies with IgG specific technology - compares to enzyme methods without the added work1
- Provides high sensitivity for clinically significant antibodies, without the noise of IgM
- Reports describe the use of Werfen products to help mitigate interference with monoclonal antibody therapies2, 3
Easy to Use.
- Save time with ready to use reagents
- No liquid screening cells to manage because antigens are precoated on the microwells
- Less hands-on time with no precision pipetting required
- Simplify training with the same methodology and procedures for all assays
Versatility and Flexibility for Labs of All Sizes.
- A wash step allows for the use of slightly lipemic or hemolyzed samples
- Run a single sample or a full batch
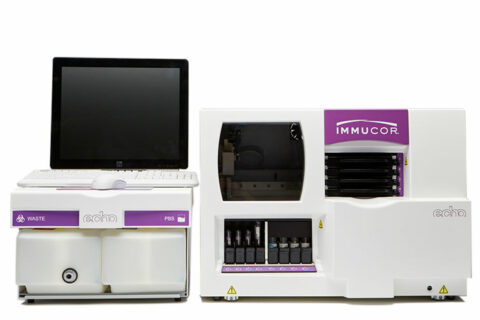
- Manual, semi-automated and fully automated scalable solutions
- Multiple options in automated red blood cell antibody detection
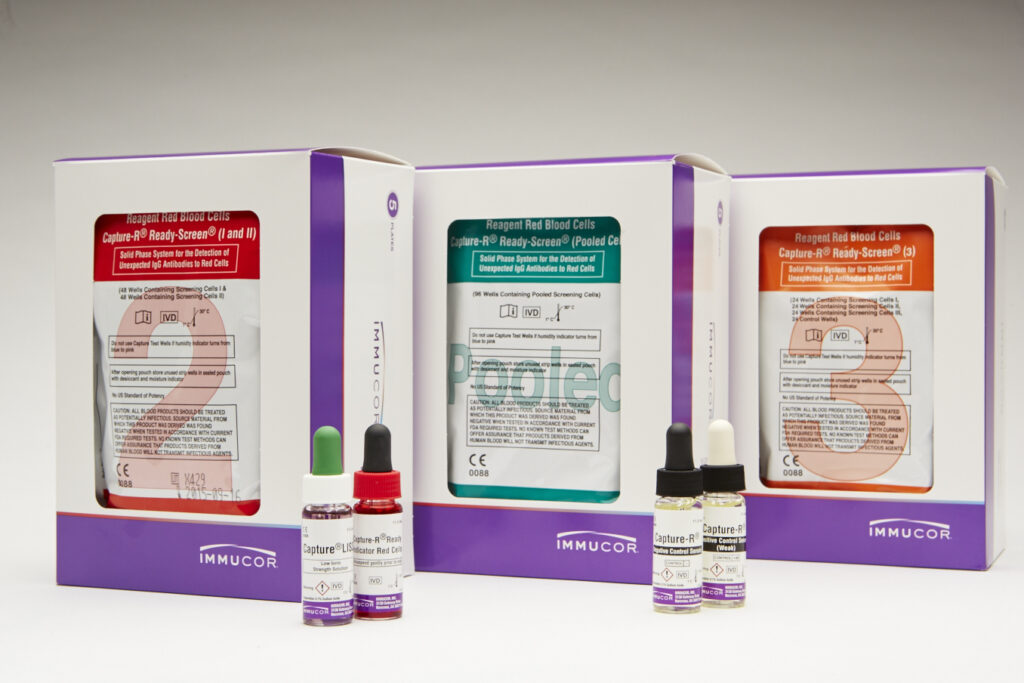
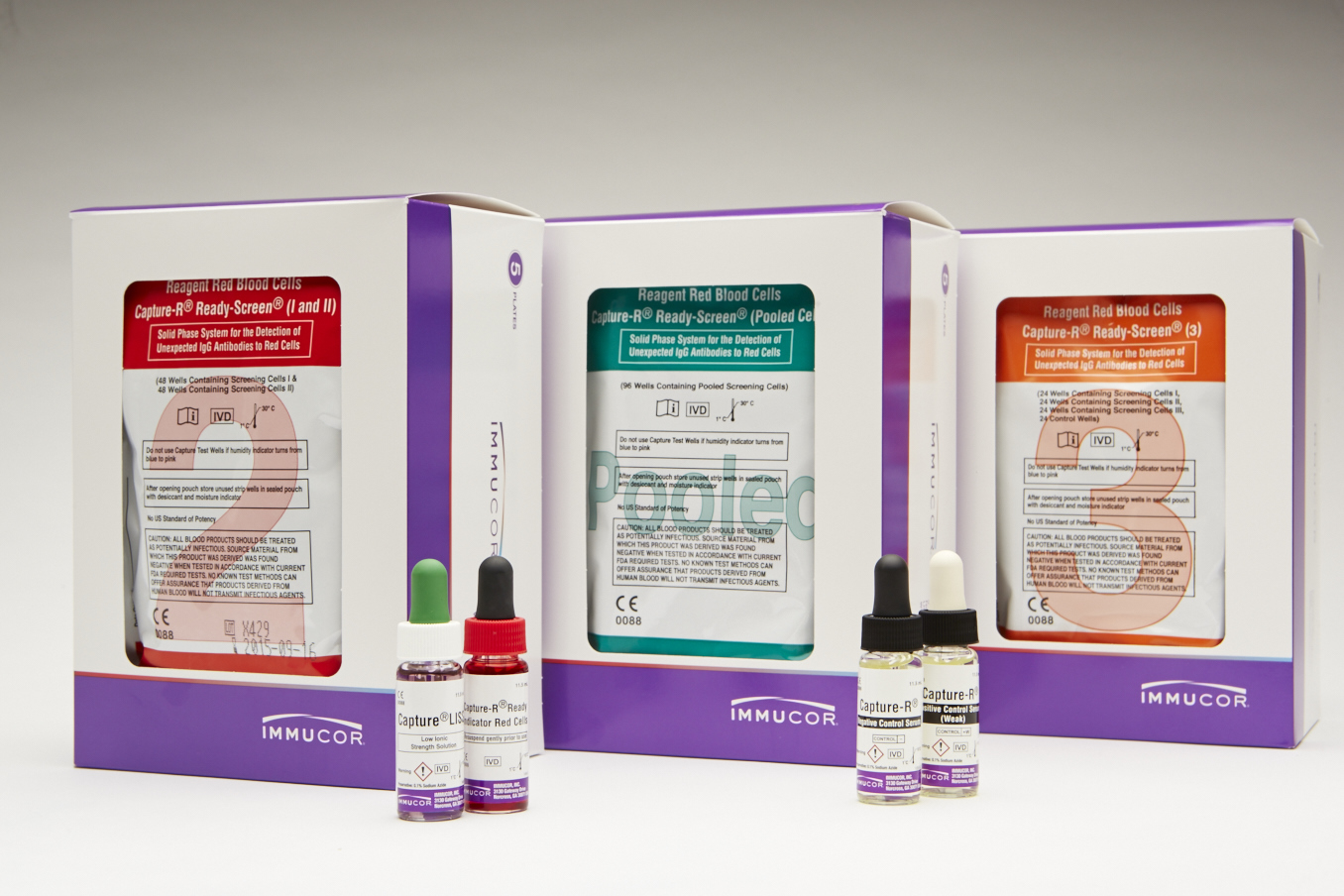
| Product | Description |
|---|---|
| Capture-R Ready-Screen (I and II) |
Contains alternating wells coated with red cell membranes from one of two group O donors. Each strip consists of eight individual wells to perform four (2-cell) antibody screens. Dia and Cw positive cells available. |
| Capture-R Ready-Screen (3) | Contains alternating wells coated with red cell membranes from one of two group O donors. Each strip consists of eight individual wells to perform two (3-cell) antibody screens. The fourth well of each antibody screen consists of a built-in positive control, a dried cell that is sensitized with IgG anti-D. Dia, Cw and Mia positive cells available. |
| Capture-R Ready-Screen (Pooled) | Each well is coated with red cell membranes prepared from a pool of two Group O donors. Each strip consists of eight individual wells to perform eight antibody screens. Dia and Cw positive cells available. |
Trust Capture for Reliability.
Capture-R Ready-Screen is the method of choice for screening over 90% of the North American blood supply and has been utilized in renowned blood donor centers globally for nearly 40 years.
Consistent and Accurate Manufacturing.
- Werfen has a rich supply of donors. Our database of over 10,000 typed donors is utilized to create the best antigen profile on every lot.
- Our donors are phenotyped by two separate laboratories using unique reagents.
1 Funato, Y. (2018, March). Scientific Study on Comparison of Column with Enzyme and Capture without Enzyme. Toyonaka, Japan; Toyonaka Hp.
2 Howard-Menk C, Crane JE, Doshi L, Papari M. HU5F9-G4 Monoclonal Anti-CD47 Therapy: A First Experience with Interference in Antibody Identification. Transfusion 2018; 58 [S2]:177A.
3 De Vooght KMK, Lozano M, Bueno JL, et al. Vox Sanguinis International Forum on typing and matching strategies in patients on anti-CD38 monoclonal therapy: summary. Vox Sanguinis 2018; 113:492-498.
Product offerings may differ by region, please check with your local Werfen representative for regulatory status in your area.
Automated Capture-R® Ready-Screen® Downloads

Resource Library
Our Certifications
Experience Our Virtual Lab
Related Products
- Testimonial
- Transfusion
- Customer
Raeann Thomas, Blood Bank Technical Supervisor, Hospital of the University of Pennsylvania, USA
Let's Get Started On Your Story- Testimonial
- Transfusion
- Customer
Kevin Elman, Transfusion Services Coordinator, North Colorado Medical Center, USA
Let's Get Started On Your Story- Testimonial
- Transfusion
- Customer
Don-Dolcetto Ngilisho, Laboratory Services Officer, Tanzania National Blood Transfusion Service
Let's Get Started On Your Story- Testimonial
- Transfusion
- Customer
Lisa Knapp, MT(ASCP), Blood Bank Coordinator, Bozeman Health Deaconess Hospital
Let's Get Started On Your Story- Testimonial
- Transfusion
- Customer
Kerry Dowling, Blood Transfusion Laboratory Manager, Southampton General Hospital University Hospital Southampton NHS Foundation Trust, United Kingdom
Let's Get Started On Your Story- Testimonial
- Transfusion
- Customer
Alexis Crisp, MLS (ASCP) CMSBB, Blood Bank Supervisor, Avera McKennan Hospital and University Health Center
Let's Get Started On Your StoryUpcoming Events,
Webinars & Workshops
Keep Up With Us