

Automated Capture-R® Ready-ID® Benefits
to Use
Specificity
Flexibility
Take your laboratory to the next level of productivity with Capture®, a proven solid phase technology for antibody screening and identification.
Capture’s IgG specific technology provides high sensitivity for clinically significant antibodies, without the noise of IgM antibodies. To improve efficiency, reagent red blood cells are bound to test wells at the time of manufacture so there are no liquid reagent red blood cells to manage.
High Accuracy.
- Detect clinically significant antibodies with IgG specific technology - compares to enzyme methods without the added work1
- Provides high sensitivity for clinically significant antibodies, without the noise of IgM
- Reports describe the use of Werfen products to help mitigate interference with monoclonal antibody therapies2, 3
Easy to Use.
- Save time with ready to use reagents
- No liquid identification-cells to manage because we use pre-coated antibody screen and identification cells
- Less hands-on time with no precision pipetting required
- Simplify training with the same methodology and procedures for all assays
Versatility and Flexibility for Labs of All Sizes.
- Run a single sample or a full batch
- Manual, semi-automated and fully automated scalable solutions
- Wide range of assays utilize the same test platform
Consistent and Accurate Manufacturing.
- Werfen has a rich supply of donors. Our database of over 10,000 typed donors is utilized to create the best antigen profile on every lot.
- Our donors are phenotyped by two separate laboratories using unique reagents.
- We apply our experience with molecular immunohematology (MIH) to enhance the value of our serological products. Each donor’s full antigen profile, including extended antigen typing by PreciseType HEA DNA analysis is provided on the Supplemental Unit Typings (SUT) with each antibody identification panel.
You have multiple options for automated identification of red blood cell antibodies:
| Product | Description |
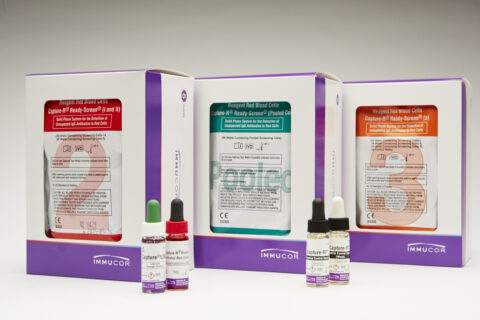
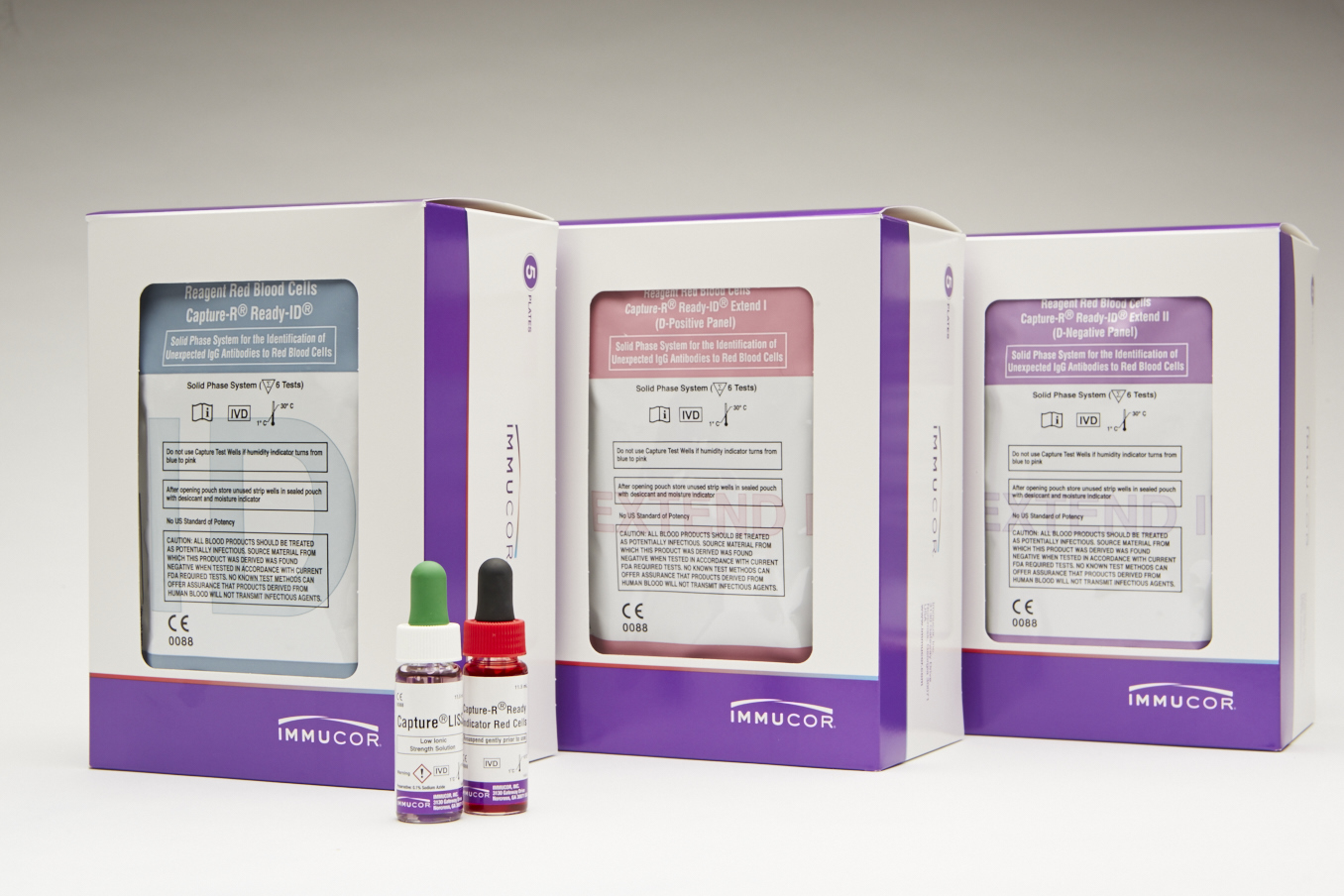
| Capture-R Ready-ID | The primary Ready-ID 2x8 strips contain test wells coated with the red cell membranes of 14 O single donors. The donors are selected to possess most of the frequently inherited antigens. |
| Capture-R Ready-ID Extend I | D positive panel: 2x8 strips contain test wells coated with D-positive red cell membranes of 14 group O single donors. |
| Capture-R Ready-ID Extend II | D negative panel: 2x8 strips contain test wells coated with D-negative red cell membranes of 13 group O single donors and 1 D-positive red cell membrane of a group O single donor. |
1 Funato, Y. (2018, March). Scientific Study on Comparison of Column with Enzyme and Capture without Enzyme. Toyonaka, Japan; Toyonaka Hp.
2 Howard-Menk C, Crane JE, Doshi L, Papari M. HU5F9-G4 Monoclonal Anti-CD47 Therapy: A First Experience with Interference in Antibody Identification. Transfusion 2018; 58 [S2]:177A.
3 De Vooght KMK, Lozano M, Bueno JL, et al. Vox Sanguinis International Forum on typing and matching strategies in patients on anti-CD38 monoclonal therapy: summary. Vox Sanguinis 2018; 113:492-498.
Product offerings may differ by region, please check with your local Werfen representative for regulatory status in your area.
Automated Capture-R® Ready-ID® Downloads

Resource Library
Our Certifications
Experience Our Virtual Lab
Related Products
- Testimonial
- Transfusion
- Customer
Raeann Thomas, Blood Bank Technical Supervisor, Hospital of the University of Pennsylvania, USA
Let's Get Started On Your Story- Testimonial
- Transfusion
- Customer
Kevin Elman, Transfusion Services Coordinator, North Colorado Medical Center, USA
Let's Get Started On Your Story- Testimonial
- Transfusion
- Customer
Don-Dolcetto Ngilisho, Laboratory Services Officer, Tanzania National Blood Transfusion Service
Let's Get Started On Your Story- Testimonial
- Transfusion
- Customer
Lisa Knapp, MT(ASCP), Blood Bank Coordinator, Bozeman Health Deaconess Hospital
Let's Get Started On Your Story- Testimonial
- Transfusion
- Customer
Kerry Dowling, Blood Transfusion Laboratory Manager, Southampton General Hospital University Hospital Southampton NHS Foundation Trust, United Kingdom
Let's Get Started On Your Story- Testimonial
- Transfusion
- Customer
Alexis Crisp, MLS (ASCP) CMSBB, Blood Bank Supervisor, Avera McKennan Hospital and University Health Center
Let's Get Started On Your StoryUpcoming Events,
Webinars & Workshops
Keep Up With Us