ImmuLINK® v2.0
A couple of editions ago we were excited to announce the launch of ImmuLINK v2.0! For those who are not familiar with ImmuLINK, it is Immucor’s software suite designed specifically to improve testing efficiency and traceability in the Blood Bank.
Some of the basic features of ImmuLINK are:
- Centralised data management
- Single user interface for all Immucor instruments
- Manage complex testing at remote sites from any location
- Provide an easier way to manage Blood Bank test data
- Customisable to fit your current or desired workflow
We have taken ImmuLINK to a whole other level with the new Panel ID Module available in v2.0. This new functionality will reduce the risk of transcription errors and will increase the traceability of all antibody detection/identification activities performed in your laboratory. Users now have the ability to verify automated assay result details auto-populated in the lot-specific Master List within the ImmuLINK user interface. A possible positive identification workflow could then look like this:
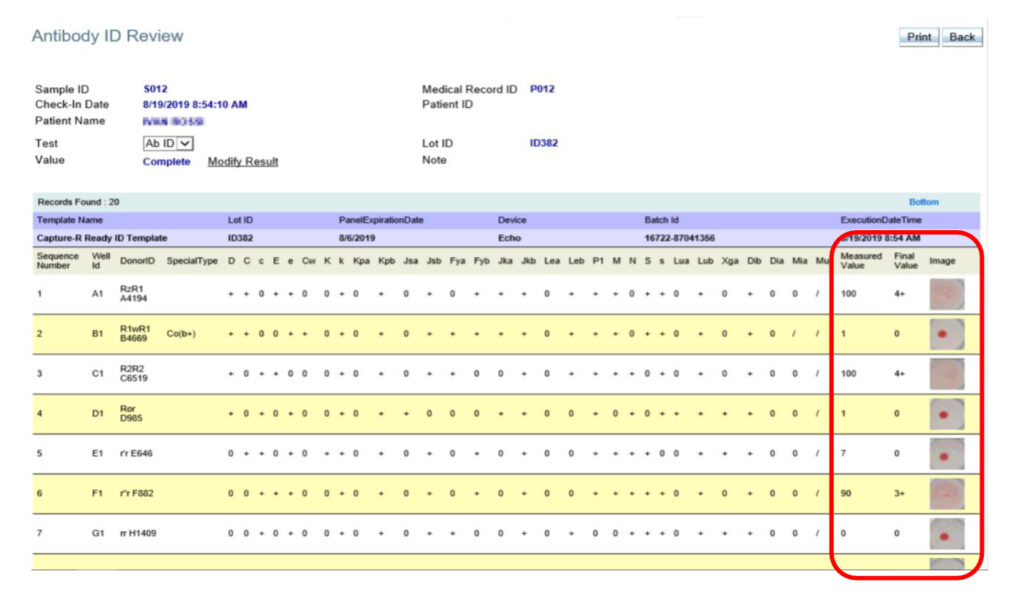
The panel result is linked through ImmuLINK to the associated Master List, based on the reagent lot identification number. Each new Immucor Master List will be automatically downloaded by ImmuLINK from the web and stored in the local ImmuLINK database. As such, you now have a single view that includes the instrument results, reaction scores, final interpretations, images and the lot-specific Master List with all the cells and reaction map.
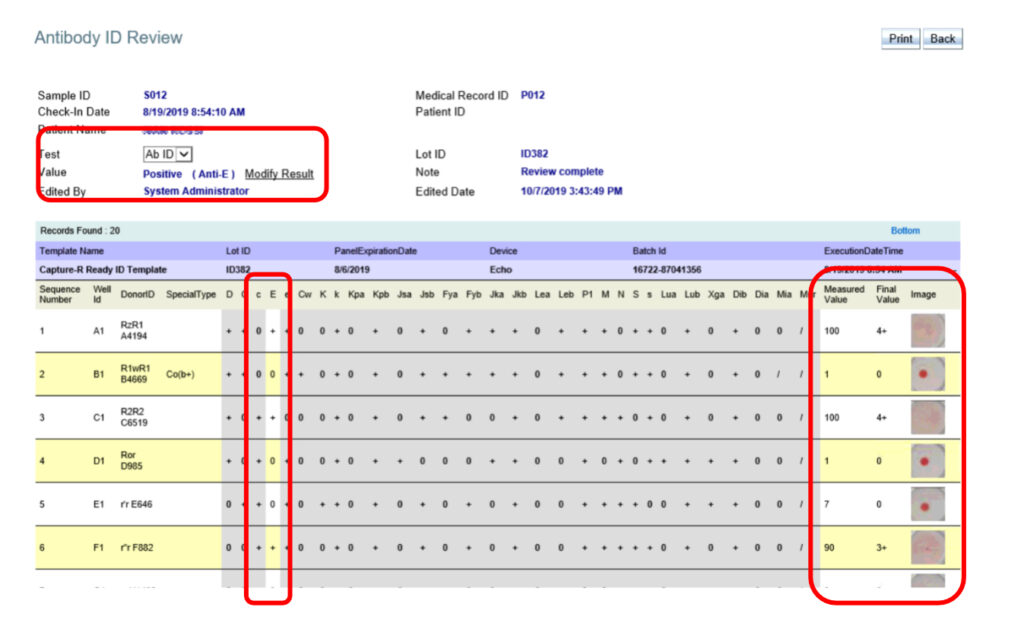
The software provides the capability to “grey out” columns associated to antibodies that can be excluded thereby allowing the authorised user to easily identify one or multiple antibodies. Multiple panels or even antibody screen results for a sample can be included in this view to have all the related information visible
at one time. The result can then be modified by the authorized user by selecting the antibodies identified.
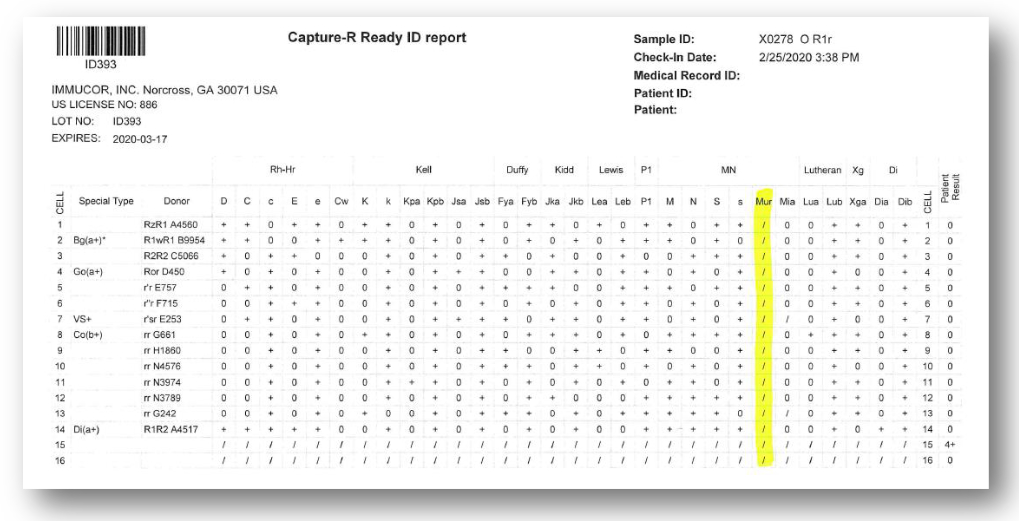
The report (above) can be printed with all the results transcribed directly into the master list. This is a key safety feature. As was described in the annual 2018 SHOT report below, errors made due to the transposition of results including antibody identification panel reactions, incorrectly transcribed from an analyser to a paper panel sheet, are a key contributor to lab errors:
ANNUAL SHOT REPORT:
Errors made due to the transposition of either samples or results during testing, and those made during result transcription continue to be recurring errors made in all elements of testing across the majority of External Quality Assurance (EQA) exercises. Although the format of an EQA exercise cannot exactly replicate clinical testing scenarios, performing checks such as sample labelling prior to the commencement of and during any serology testing, and checking results prior to any manual reporting step, could be considered to be routine practice across all laboratories. In seven exercises (17E6, 17R8, 17R10, 18R2, 18E3, 18R5 and 18R8) a number of laboratories made procedural errors which have the potential to occur
during testing or recording results of ABO typing, antibody screening and identification, crossmatching and phenotyping. Causes reported include antibody identification panel reactions incorrectly transcribed from an analyser to paper panel sheet, switching results of phenotyping at data entry, inadvertently testing sample from a previous UK NEQAS exercise, and testing Patient 1 or 3 twice in place of Patient 2.

Total of 150 lab errors (51 near misses of HTR) that continue to put patients at risk. (SHOT report 2018)
If you would like a demo of our ImmuLINK v2.0 software, please contact your Immucor sales representative!



